Data
Access AlphaStreet’s high quality, time-sensitive and accurate information that will help you find Alpha. Our API’s simplify how you can partake and use the information.
High
quality
time-sensitive and accurate
financial data to find Alpha
Audio
synced with Live and Archived
transcript streaming
Institutional
quality data
vetted by our experienced
analysts
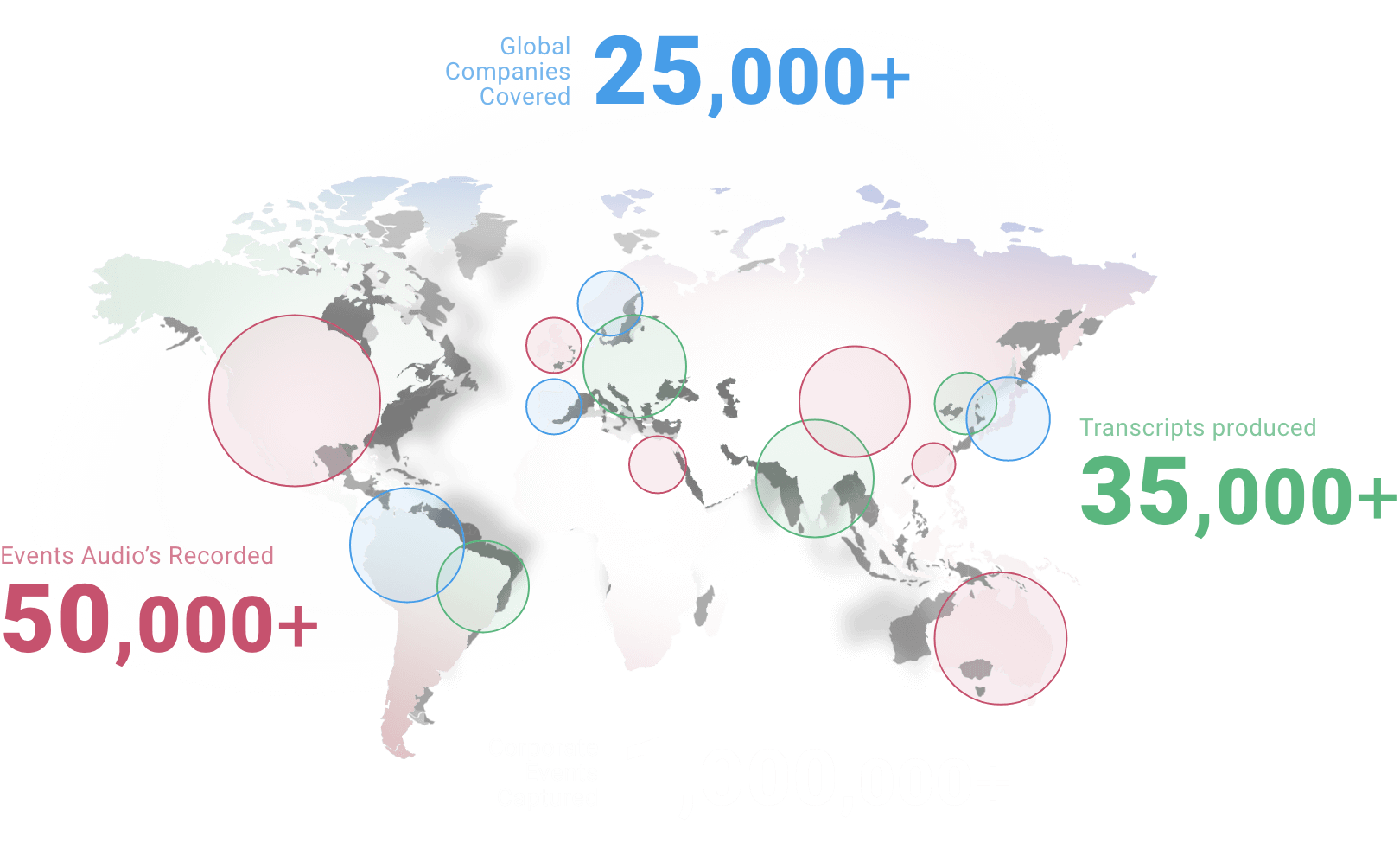
Global
coverage
one stop hub
Events & Corporate
Actions Calendar
Industry's most accurate corporate events data, powered by AI.
40+
event types tracked
AI/NLP
friendly metadata structure
Extensive
historical data
API
delivery
Transcripts
World’s largest and most accurate earnings
and other conference call transcript
Live streaming
Smart tagged
Industry best TAT
Audio synced
AlphaGraphs
Visually compelling and easy to digest for
insights into companies, competition and
industries.
Research
Unbiased research for coverage initiation or generating
investment ideas. Combine our research with our data
to get insights to make informed decisions.

CxO & Experts
interviews
If you are a CxO, tell the investors your story or if
you are a subject matter expert(SME), share your
expertise.


Who uses us

Global public companies

Hedge funds and Quant Funds

Portfolio managers and Asset managers

Financial Institutions

Business intelligence firms












